What is the rate constant of iodine clock reaction?
Just so, what is the rate order of iodine clock reaction?
The thiosulfate ions quickly consume the triiodide ions. The blue-black color does not appear until all of the thiosulfate ions are consumed. The thiosulfate ions are the limiting reactant. The rate of reaction is first-order in potassium iodine.
Likewise, what is the rate constant of a reaction? The rate constant, k, is a proportionality constant that indicates the relationship between the molar concentration of reactants and the rate of a chemical reaction. The rate constant may be found experimentally, using the molar concentrations of the reactants and the order of reaction.
Keeping this in view, what are the kinetics of an iodine clock?
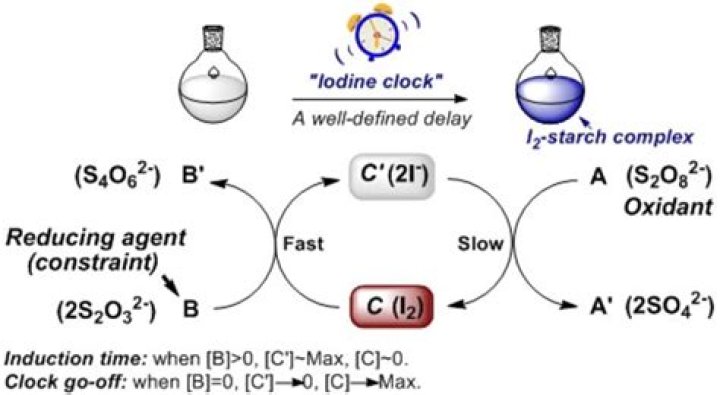
The “clock reaction” is a reaction famous for its dramatic colorless-to-blue color change, and is often used in chemistry courses to explore the rate at which reactions take place. The color change occurs when I2 reacts with starch to form a dark blue iodine/starch complex.
What type of reaction is the iodine clock reaction?
There are actually two chemical reactions going on at the same time when you combine the solutions. During these reactions two forms of iodine created – the elemental form and the ion form. In Reaction # 1 iodide ions react with hydrogen peroxide to produce iodine element which is blue in the presence of starch.
Related Question Answers
What is the concentration of iodine?
The iodine-replete healthy adult has about 15–20 mg of iodine, 70%–80% of which is contained in the thyroid [6]. Median urinary iodine concentrations of 100–199 mcg/L in children and adults, 150–249 mcg/L in pregnant women and >100 mcg/L in lactating women indicate iodine intakes are adequate [3].Why is iodine not in the rate equation?
Experimental data shows the reaction to be first order with respect to both propanone and hydrogen ions, but zero order for iodine. The reactant iodine is not in the rate equation at all but hydrogen ions are present, despite not being reactants.Why is the iodine clock reaction important?
Purpose: The rate law for the reaction of an “iodine clock” reaction is to be established. When the iodine concentration increases, it reacts with the starch in the solution to form a complex, turning it a deep blue-black color.How does temperature affect iodine clock reaction?
The reaction known as the Iodine Clock reaction was used to study the rates of reaction. A catalyst for the reaction (Copper II Sulfate) was added to solution A, solution B, and both solutions and the rate of the reaction was recorded. Results. As temperature went up the rate of reaction increased.What is the activation energy of iodine clock reaction?
Iodine-Clock Activation Energy(1/T) for the iodine clock reaction should reveal a slope of about -6230. Thus, (-Ea/R) = -6230. Using an ideal gas constant of R = 8.314 J/K. mol gives Ea = 6800 * 8.314 = 51,800 J/mol, or 51.8 kJ/mol.
What happens when sodium thiosulphate reacts with iodine?
Sodium thiosulfate react with iodine to produce tetrathionate sodium and sodium iodide. Sodium thiosulfate - diluted solution.How does the iodine clock reaction work?
This clock reaction uses sodium, potassium or ammonium persulfate to oxidize iodide ions to iodine. Sodium thiosulfate is used to reduce iodine back to iodide before the iodine can complex with the starch to form the characteristic blue-black color.What are rate constant units?
k is the first-order rate constant, which has units of 1/s. The method of determining the order of a reaction is known as the method of initial rates. The overall order of a reaction is the sum of all the exponents of the concentration terms in the rate equation.How do you calculate initial rate of reaction?
The initial rate is equal to the negative of the slope of the curve of reactant concentration versus time at t = 0.How do you determine reaction order?
The overall order of the reaction is found by adding up the individual orders. For example, if the reaction is first order with respect to both A and B (a = 1 and b = 1), the overall order is 2. We call this an overall second order reaction.How do you calculate the average rate constant?
Plan The average rate is given by the change in concentration, ∆[A], divided by the change in time, ∆t. Because A is a reactant, a minus sign is used in the calculation to make the rate a positive quantity.Is the iodine clock reaction exothermic or endothermic?
In terms of thermodynamics, this reaction is exothermic and should occur spontaneously.How do you calculate activation energy?
NOW, Activation Energy:So now we can use it to calculate the Activation Energy by graphing lnk versus 1/T. When the lnk (rate constant) is plotted versus the inverse of the temperature (kelvin), the slope is a straight line. The value of the slope (m) is equal to -Ea/R where R is a constant equal to 8.314 J/mol-K.
How do you write a rate law?
A rate law relates the concentration of the reactants to the reaction rate in a mathematical expression. It is written in the form rate = k[reactant1][reactant2], where k is a rate constant specific to the reaction. The concentrations of the reactants may be raised to an exponent (typically first or second power).Is Iodine a hazardous chemical?
Higher exposures may cause a build-up of fluid in the lungs (pulmonary edema), a medical emergency, with severe shortness of breath. ? Iodine can cause headache, metallic taste, nausea, vomiting, diarrhea and abdominal pain.What is the catalyst in the iodine clock reaction?
Note: Sulfuric acid is a catalyst for this reaction.Why is it called iodine clock reaction?
The iodine clock reaction is a favorite demonstration reaction in chemistry classes. The reaction is called a clock reaction because the amount of time that elapses before the solution turns blue depends on the concentrations of the starting chemicals.What is the role of starch in the iodine clock reactions?
Potassium persulphate is used to oxidize iodide ions to iodine, in the presence of starch and a small amount of thiosulphate ions. When the thiosulphate is exhausted (by reaction with the iodine produced), the dark blue iodine-starch complex is formed.Which species is responsible for the blue color that appears during the iodine clock reaction?
Chemical Test for Starch or IodineAmylose in starch is responsible for the formation of a deep blue color in the presence of iodine.
Why was the total volume of solution A kept constant in the iodine clock demonstration?
The total volume was kept constant in order to keep consistency across the calculations of concentration for each of the different trials. -In order to investigate the effect of KIO3 concentration on the rate of the iodine clock reaction, the composition and amount of Solution B was not changed.What are the 4 factors that affect the rate of reaction?
There are four main factors that can affect the reaction rate of a chemical reaction:- Reactant concentration. Increasing the concentration of one or more reactants will often increase the rate of reaction.
- Physical state of the reactants and surface area.
- Temperature.
- Presence of a catalyst.



