Overview. The term ATPase applies to the activity of any enzyme's ability to decompose ATP, including metabolic enzymes involved in anabolic processes that need energy, as well as enzymes promoting transport across membrane. This process of ATP synthesis is often called oxidative phosphorylation. Simply so, what does an ATPase do?
Overview. The term ATPase applies to the activity of any enzyme's ability to decompose ATP, including metabolic enzymes involved in anabolic processes that need energy, as well as enzymes promoting transport across membrane.
Also, what is myosin ATPase activity? showSearch. Myosin ATPase (EC 3.6.4.1) is an enzyme with systematic name ATP phosphohydrolase (actin-translocating). This enzyme catalyses the following chemical reaction ATP + H2O ADP + phosphate. ATP hydrolysis provides energy for actomyosin contraction.
Just so, what protein has ATPase activity?
For example, in both prokaryotes and eukaryotes, another type of chaperone, DnaJ, stimulates the ATPase activity of Hsp70s by accelerating the rate of ATP hydrolysis (11, 12, 13). In eukaryotes, a protein called Hip has been identified that stabilizes the ADP bound form by inhibiting the release of nucleotide (14).
Does actin have ATPase activity?
Actin is an ATPase, which means that it is an enzyme that hydrolyzes ATP. This group of enzymes is characterised by their slow reaction rates. It is known that this ATPase is “active”, that is, its speed increases by some 40,000 times when the actin forms part of a filament.
Related Question Answers
Where is ATPase used?
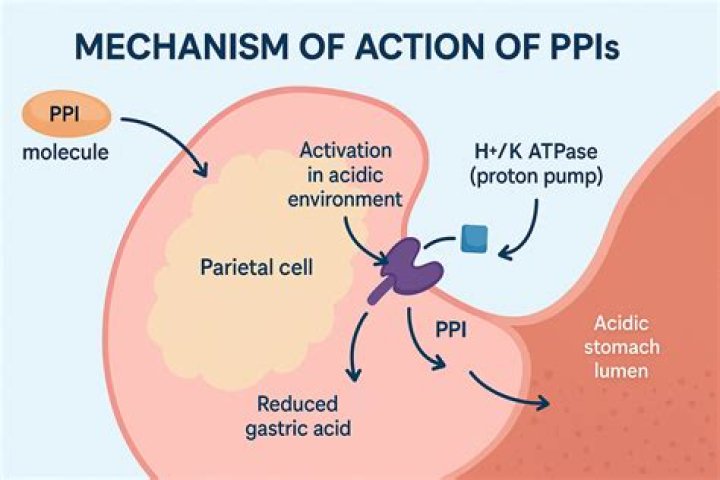
Proton ATPases are used by cells to transport protons against the electrochemical gradient by harnessing the energy of ATP hydrolysis. Because ATP is hydrolyzed, the type of transport is referred to as active transport. One use of a proton ATPase is to acidify a cellular compartment, such as a lysosome. What is the function of the Na +/ K+ pump?
The sodium-potassium pump system moves sodium and potassium ions against large concentration gradients. It moves two potassium ions into the cell where potassium levels are high, and pumps three sodium ions out of the cell and into the extracellular fluid. Is ATPase reversible?
Reversible ATPases that can use a proton gradient to synthesize ATP or create a proton gradient upon ATP hydrolysis. They are found in bacteria, plants (chloroplasts) and eukaryotes (mitochondria). What happen if ATPase would be inhibited?
The inhibition of the Na/K pump will allow Na ions to accumulate in the cell, as K ion will fall. So if the Na/K pump was inhibited and stops working, then many functional problems will occur in the cell. Na ion concentration will accumulate within the cell and intracellular K ion concentration falls. Is ATPase a carrier protein?
Some examples of pumps for active transport are Na+-K+ ATPase , which carries sodium and potassium ions, and H+-K+ ATPase, which carries hydrogen and potassium ions. Both of these are antiporter carrier proteins. What is the substrate for ATPase?
Thus, there exists a consensus that a metal-ATP complex is the substrate for all three ATPases. What cells have sodium potassium pumps?
also known as the Na+/K+ pump or Na+/K+-ATPase, this is a protein pump found in the cell membrane of neurons (and other animal cells). It acts to transport sodium and potassium ions across the cell membrane in a ratio of 3 sodium ions out for every 2 potassium ions brought in. Is ATPase active transport?
There are two mechanisms on the surface membrane of the cells that pump out Ca2+ ions: a plasma membrane Ca-ATPase (PMCA) and a Na+–Ca2+ exchanger (NCX). The PMCA couples uphill Ca2+ transport to the chemical energy in ATP hydrolysis, and thus it is a primary active transport mechanism. Do myosin heads have ATPase activity?
In all myosins, the head domain is a specialized ATPase that is able to couple the hydrolysis of ATP with motion. A critical feature of the myosin ATPase activity is that it is actin-activated. When packed tightly together in a thick filament, many myosin head domains can interact simultaneously with actin filaments. What kind of protein is ATP synthase?
The mitochondrial ATP synthase is a multimeric enzyme complex with an overall molecular weight of about 600,000 Da. The ATP synthase is a molecular motor composed of two separable parts: F1 and Fo. The F1 portion contains the catalytic sites for ATP synthesis and protrudes into the mitochondrial matrix. Is GTPase a phosphatase?
But GTPase is both a hydrolase and phosphatase. A Hydrolase doesn't always remove a phosphate, but in this case it does. A phosphatase is a type of hydrolase, though. "Hydrolases are classified as EC 3 in the EC number classification of enzymes. Is ATPase primary or secondary active transport?
Because the transport process uses ATP as an energy source, it is considered an example of primary active transport. Where is ATPase found in mitochondria?
Human mitochondrial (mt) ATP synthase, or complex V consists of two functional domains: F1, situated in the mitochondrial matrix, and Fo, located in the inner mitochondrial membrane. Complex V uses the energy created by the proton electrochemical gradient to phosphorylate ADP to ATP. What are ATP synthase particles?
The ATP synthase is a mitochondrial enzyme localized in the inner membrane, where it catalyzes the synthesis of ATP from ADP and phosphate, driven by a flux of protons across a gradient generated by electron transfer from the proton chemically positive to the negative side. Are ATPase and ATP synthase the same?
ATPase enzyme is commonly linked with the proton pump. It helps to breakdown ATP into ADP and inorganic Phosphate. On the other hand, ATP synthase is an enzyme used to synthesize ATP from using ADP and inorganic phosphate. Is dynein an ATPase?
Dyneins are large molecular motors that hydrolyze ATP to generate a minus-end-directed force along microtubules. Each dynein consists of one to three dynein heavy chains (HCs), which encompass the ATPase activity, complexed to intermediate (IC), light-intermediate (LIC), and light chains (LC). What is an ATPase inhibitor?
Description. ATP synthase inhibitor prevents the enzyme from switching to ATP hydrolysis during collapse of the electrochemical gradient, for example during oxygen deprivation. ATP synthase inhibitor forms a one to one complex with the F1 ATPase, possibly by binding at the alpha-beta interface. Is myosin thick or thin?
The thick filament, myosin, has a double-headed structure, with the heads positioned at opposite ends of the molecule. During muscle contraction, the heads of the myosin filaments attach to oppositely oriented thin filaments, actin, and pull them past one another. Which part of myosin has ATPase activity?
The enzyme at the binding site on myosin is called ATPase. The energy released during ATP hydrolysis changes the angle of the myosin head into a “cocked” position. The myosin head is then in a position for further movement, possessing potential energy, but ADP and Pi are still attached. How does ATP cause muscle contraction?
ATP then binds to myosin, moving the myosin to its high-energy state, releasing the myosin head from the actin active site. ATP can then attach to myosin, which allows the cross-bridge cycle to start again; further muscle contraction can occur. What are Myofibrils?
Myofibril, very fine contractile fibres, groups of which extend in parallel columns along the length of striated muscle fibres. The myofibrils are made up of thick and thin myofilaments, which help give the muscle its striped appearance. What is the sarcomere?
A sarcomere is the basic contractile unit of muscle fiber. Each sarcomere is composed of two main protein filaments—actin and myosin—which are the active structures responsible for muscular contraction. The most popular model that describes muscular contraction is called the sliding filament theory. Why do warm up exercises increase myosin ATPase activity?
Myosin ATPase is an enzyme that is involved in muscle contraction. Athletes do warm- up exercises prior to athletic performance. Warm-up exercises increase myosin-ATPase activity because of the contraction of the muscles, which provides hydrolysis and energy . What is myosin heavy chain?
Myosin heavy chain (MHC) is the motor protein of muscle thick filaments. Most organisms produce many muscle MHC isoforms with temporally and spatially regulated expression patterns. Is kinesin an ATPase?
Kinesins move along microtubule (MT) filaments, and are powered by the hydrolysis of adenosine triphosphate (ATP) (thus kinesins are ATPases), a type of enzyme. What is the function of at tubule?
The function of T-TUBULES is to conduct impulses from the surface of the cell (SARCOLEMMA) down into the cell and, specifically, to another structure in the cell called the SARCOPLASMIC RETICULUM. Is Actin a thick or thin filament?
Your muscles work in a similar fashion. Muscles are composed of two major protein filaments: a thick filament composed of the protein myosin and a thin filament composed of the protein actin. What is the difference between myosin and actin?
The main difference between actin and myosin is that actin is a protein that produces thin contractile filaments within muscle cells, whereas myosin is a protein that produces the dense contractile filaments within muscle cells. Who discovered actin?
Straub
What are the principal proteins of muscle contraction?
Tropomyosin and troponin are two main proteins that regulate muscle contraction and relaxation (Choi and Kim, 2009; Zot and Potter, 1987). They prevent the activation of actomyosin ATPase in the absence of calcium ions by interacting with actin filaments to block the myosin binding site. How is myosin activated?
MLCK is activated by calmodulin in response to an increase in intracellular calcium. It then goes on to phosphorylate regulatory myosin light chains at residues serine 19 and threonine 18 [2][3]. These phosphorylations enhance the ATPase activity of actin-activated myosin and so promotes myosin-driven contraction. Where is F actin found?
Filamentous actin (F-actin) units are also referred to as microfilaments and are highly conserved, proteinous components found near ubiquitously in eukaryotic cytoskeletons. F-actin and other actin proteins generally have structural roles in cells. How many types of myosin are there?
Three types of unconventional myosins predominate: myosin I, myosin V, and myosin VI. The unconventional myosin I and V categories contain multiple members. Is actin a globular protein?
Actin is a globular protein that exists in the monomeric form (G-actin) and polymeric filamentous form (F-actin). G-actin is an ATPase, containing a deep cleft where the adenosine nucleotide binds in the presence of divalent magnesium. What role does actin play in muscle contraction?
Muscle contraction thus results from an interaction between the actin and myosin filaments that generates their movement relative to one another. The molecular basis for this interaction is the binding of myosin to actin filaments, allowing myosin to function as a motor that drives filament sliding.