What are the components of mass spectrometer?
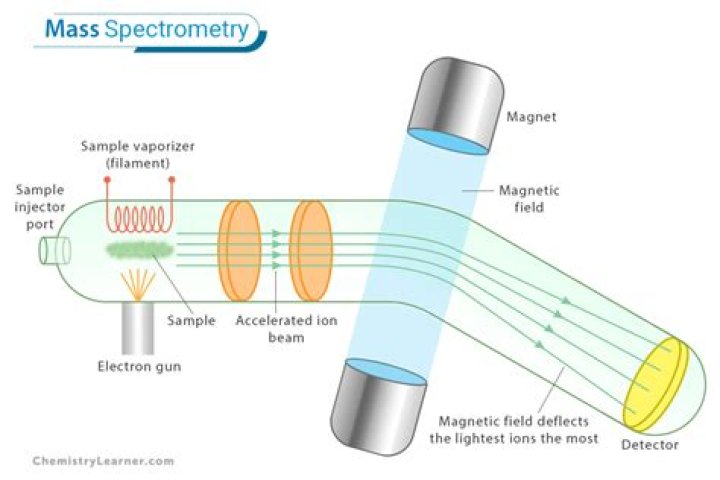
- Inlet system (LC, GC, Direct probe etc)
- Ion source (EI, CI, ESI, APCI, MALDI, etc)
- Mass analyzer (Quadrupole, TOF, Ion Trap, Magnetic Sector)
- Detector (Electron Multiplier, Micro Channel Plates MCPs)
Keeping this in view, which of the following is not a component of mass spectrometer?
Explanation: Sweep generator is not a component of a mass spectrometer. It is a component of NMR spectrometer.
Similarly, what is the principle of mass spectroscopy? Basic Principle
A mass spectrometer generates multiple ions from the sample under investigation, it then separates them according to their specific mass-to-charge ratio (m/z), and then records the relative abundance of each ion type.
Also, what are the four stages of a mass spectrometry?
There are four stages in a mass spectrometer which we need to consider, these are – ionisation, acceleration, deflection, and detection.
Where is mass spectrometry used?
Specific applications of mass spectrometry include drug testing and discovery, food contamination detection, pesticide residue analysis, isotope ratio determination, protein identification, and carbon dating.
Related Question Answers
What are the three major components of a mass spectrometer?
A mass spectrometer consists of three components: an ion source, a mass analyzer, and a detector. The ionizer converts a portion of the sample into ions.How does mass spectrometer work?
A mass spectrometer can measure the mass of a molecule only after it converts the molecule to a gas-phase ion. To do so, it imparts an electrical charge to molecules and converts the resultant flux of electrically charged ions into a proportional electrical current that a data system then reads.Who discovered the mass spectrometer?
Wilhelm WienWhat are the applications of mass spectroscopy?
Specific applications of mass spectrometry include drug testing and discovery, food contamination detection, pesticide residue analysis, isotope ratio determination, protein identification, and carbon dating.How is abundance measured in a mass spectrometer?
The relative abundance for a specific ion in the sample can be calculated by dividing by the number of ions with a particular m / z m/z m/z ratio by the total number of ions detected. At the end of the experiment, the instrument generates a mass spectrum for the sample, which plots relative abundance vs. m/z .What are the advantages of mass spectrometry?
Mass spectrometry (MS) has become the major analytic technology, with the biggest market share and the fastest growth rate as it offers important advantages, namely, sensitivity and specificity of detection, analyte structural information, and (potentially) unambiguous detection and quantitation of analytes.Why pressure is kept low in mass spectrometer?
Mass spectrometers require a low operating pressure in order to avoid high voltage discharges and to ensure that the mean free path of the ions is long enough to avoid collisions with the background atmosphere in the system.What is positive and negative mode in Mass Spectrometry?
In the positive ion mode protonated and/or alkali adduct analyte molecules generally observed in the mass spectra. In the negative ion mode operation peaks corresponding to deprotonated analyte molecules are observed. ESI allows production of multiply charged ions.What does M Z mean?
mass-to-charge ratioWhy is the mass spectrometer kept under vacuum?
Ions are created in the ion source. If a high vacuum provides a long mean free path exceeding the dimension of the chamber, ions can easily reach the detector. QPMS is usually operated at around 10-3 to 10-4 Pa pressure, which corresponds to 5m to 50m of a mean free path.Why do ions formed in a mass spectrometer have a positive charge?
It's important that the ions produced in the ionisation chamber have a free run through the machine without hitting air molecules. The vaporised sample passes into the ionisation chamber. The electrically heated metal coil gives off electrons which are attracted to the electron trap which is a positively charged plate.Why is the sample ionised in a mass spectrometer?
Because a mass spectrometer works by moving the particles it's examining with electric and magnetic fields. If a particle is not ionised then it has zero charge and it's mass to charge ratio is identical to all other uncharged particles - so you cannot determine its mass.What do you mean by mass spectrograph?
mass spectrograph. A device that uses electrical or magnetic fields to determine the masses of atoms or molecules in a sample. A beam of ions is passed through the electrical or magnetic field.Is mass spectrometry quantitative or qualitative?
MS enables direct identification of molecules based on the mass-to-charge ratio as well as fragmentation patterns. Thus, it fulfils the role of a qualitative analytical technique with high selectivity.What is mass spectrometry PPT?
INTRODUCTION: ?Mass spectrometry is a powerful analytical technique used to quantify known materials, to identify unknown compounds within a sample and to elucidate the structure and chemical properties of different molecules.What is difference between mass spectroscopy and mass spectrometry?
Spectroscopy refers to the study of how radiated energy and matter interact. The energy is absorbed by the matter, creating an excited state. Spectrometry is the application of spectroscopy so that there are quantifiable results that can then be assessed.What does intensity mean in mass spectrometry?
Relative abundanceWhy is mass spectrometry better than other techniques?
Mass spectrometry is sensible and preciseCoupled to liquid chromatography (LC), LC-MS can deliver highly precise quantification. Also, when performed by experts, it is highly reproducible and accurate over several orders of magnitude (of course, this is molecule-dependant!).
What is Cone voltage in mass spectrometry?
Cone voltage is an obvious answer if you lose the ions in the source , collision energy if you lose them during MS/MS. In my experience source parameters like temperature and gases can also be parameters to evaluate if you have "fragile ions".When was mass spectrometer invented?
1912What principle is used in a mass spectrograph to estimate the mass of a charged particle?
The basic principle on which mass spectrometry operates is that a stream of charged particles is deflected by a magnetic field. The amount of the deflection depends on the mass and the charge on the particles in the stream.What are the limitations of mass spectrometry?
While GC-MS is a powerful technique there are some limitations that are not often mention in these media presentations. Two such limitations are the compounds have to be in a database and be volatile. To get around the volatility issue, Liquid chromatography mass spectrometry (LC-MS) has increased in popularity.How do you do mass spectrometry?
Mass Spectrometry- A small sample is ionized, usually to cations by loss of an electron. The Ion Source.
- The ions are sorted and separated according to their mass and charge. The Mass Analyzer.
- The separated ions are then measured, and the results displayed on a chart. The Detector.



