What are some common pH indicators?
Keeping this in view, what are the different types of pH indicators?
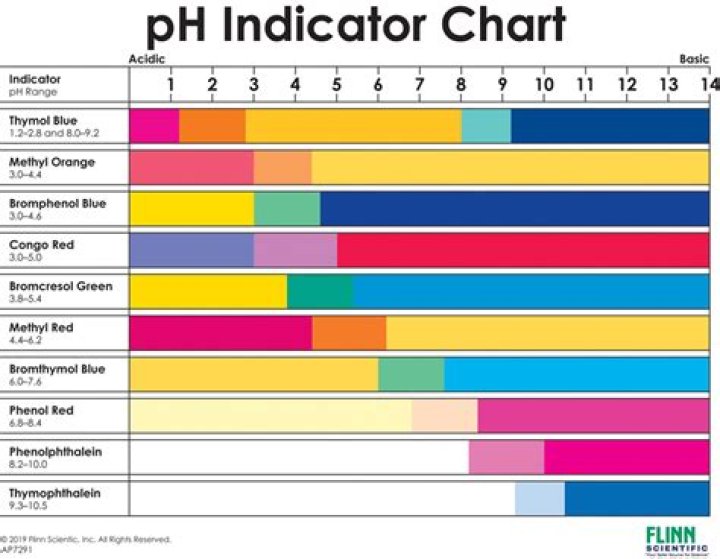
Common Acid-Base Indicators Tried-and-true indicators include thymol blue, tropeolin OO, methyl yellow, methyl orange, bromphenol blue, bromcresol green, methyl red, bromthymol blue, phenol red, neutral red, phenolphthalein, thymolphthalein, alizarin yellow, tropeolin O, nitramine, and trinitrobenzoic acid.
Secondly, what are common indicators? Common indicators include:
- Litmus: A dye extracted from various species of lichen that changes colour around the neutral range.
- Phenolphthalein: A synthetic indicator that changes colour in the basic range.
- Methyl orange: A synthetic indicator that changes colour in the acidic range.
Secondly, what are common uses of pH indicators?
pH indicators are used to give a rough value of pH of a chemical solution. For precise measurements, a pH meter is used. Alternatively, absorbance spectroscopy may be used with a pH indicator to calculate the pH using Beer's law.
What are examples of pH indicators?
Because acidity and alkalinity relate to pH, they may also be known as pH indicators. Examples of acid-base indicators include litmus paper, phenolphthalein, and red cabbage juice.
Related Question Answers
Which gives a better measure of pH?
It mostly depends on the observer how good he is in differentiating between the colours ,while the pH-meter gives us the exact value of the pH and we don't need to look for anything else.So,pH-meter is a better choice among these two methods.What are the three types of indicators?
There are three types of economic indicators, depending on their timing: leading, lagging, and coincident indicators. Leading indicators signal changes before the economy as a whole changes.What are the two types of indicators?
indicators have six major types. they are the organic compounds. They have different colours in acidic and alkaline solutions. Litmus is a common indicator.Types of Indicators with Uses and Examples in Chemistry
- thymol blue.
- mromthymol blue.
- methyl orange.
- phenolphthalein.
- litmus.
- bromcresol green.
What are the four types of indicators?
According to this typology, there are four types of indicators: input, output, outcome and impact.What is the best pH indicator?
Some of the most widely-used pH testing tools are pH indicators, including phenolphthalein (range pH 8.2 to 10.0; colorless to pink), bromthymol blue (range pH 6.0 to 7.6; yellow to blue), and litmus (range pH 4.5 to 8.3; red to blue).What are three natural indicators?
Some examples of natural indicators are red cabbage, turmeric, grape juice, turnip skin, curry powder, cherries, beetroots, onion, tomato, etc. Some flowers like hydrangeas can determine the acidity or basicity of the soil.What are different types of indicators?
Although they may all be used for pH detection they are not all suitable for the same purpose.- Universal indicator.
- Litmus.
- Phenolphthalein.
- Methyl orange.
- Bromothymol blue.
- Choice of indicator summary.
What is a natural pH indicator?
The natural world has given us numerous plants, from beets to grapes to onions, that can be used to test the pH levels of a solution. These natural pH indicators include: Cherries: Cherries and their juice are red in an acidic solution, but they turn blue to purple in a basic solution.Do indicators affect pH?
When used as a dilute solution, a pH indicator does not have a significant impact on the acidity or alkalinity of a chemical solution. The principle behind the function of an indicator is that it reacts with water to form the hydrogen cation H+ or hydronium ion H3O+. pH indicators are usually weak acids or weak bases.What are examples of pH?
What does it mean for a solution to be acidic or basic (alkaline)?| pH Value | H+ Concentration Relative to Pure Water | Example |
|---|---|---|
| 0 | 10 000 000 | battery acid |
| 1 | 1 000 000 | gastric acid |
| 2 | 100 000 | lemon juice, vinegar |
| 3 | 10 000 | orange juice, soda |
How is pH defined?
pH is a measure of hydrogen ion concentration, a measure of the acidity or alkalinity of a solution. The pH scale usually ranges from 0 to 14. Aqueous solutions at 25°C with a pH less than 7 are acidic, while those with a pH greater than 7 are basic or alkaline.How does a pH indicator work?
HOW DO pH INDICATORS WORK? pH indicators detect the presence of H+ and OH-. They do this by reacting with H+ and OH-: they are themselves weak acids and bases. If an indicator is a weak acid and is coloured and its conjugate base has a different colour, deprotonation causes a colour change.Why are indicators used?
The common application of indicators is the detection of end points of titrations. The colour of an indicator alters when the acidity or the oxidizing strength of the solution, or the concentration of a certain chemical species, reaches a critical range of values.What are indicators with examples?
Acid-base indicators are chemicals used to determine whether an aqueous solution is acidic, neutral, or alkaline. Because acidity and alkalinity relate to pH, they may also be known as pH indicators. Examples of acid-base indicators include litmus paper, phenolphthalein, and red cabbage juice.What is the pH of universal indicator?
Universal indicator is a mixture of dyes that changes colour gradually over a range of pH from 4-14. The colour is used to indicate pH directly (1). The main components of a Universal indicator, in the form of a solution, are thymol blue, methyl red, bromothymol blue and phenolphthalein.Where are indicators used in everyday life?
Everyday uses of indicators include: Testing the acidity or basicity of soils, as some plants grow best in slightly acidic soils (such as azaleas and pineapples) while others grow best in slightly basic soils (such as violets):What are indicators give example?
Examples of acid-base indicators include litmus paper, phenolphthalein, and red cabbage juice. An acid-base indicator is a weak acid or weak base that dissociates in water to yield the weak acid and its conjugate base or else the weak base and its conjugate acid.What are the dashboard symbols?
What 15 Symbols On Your Car's Dashboard Mean- Engine Temperature Warning Light. iStock.
- Tire Pressure Warning Light. iStock.
- Oil Pressure Warning. iStock.
- Traction Control. iStock.
- Engine Warning. iStock.
- Antilock Brake Warning. iStock.
- Automatic Shift Lock or Engine Start Indicator. iStock.
- Battery Alert. iStock.
What is the most common indicator?
The litmus paper is the most commonly used indicator in laboratory- Litmus paper is made by the chemical substance called litmus that is extracted from lichens.
- The solution of litmus contains some dyes which are absorbed on the filter paper and by this way litmus papers are made.