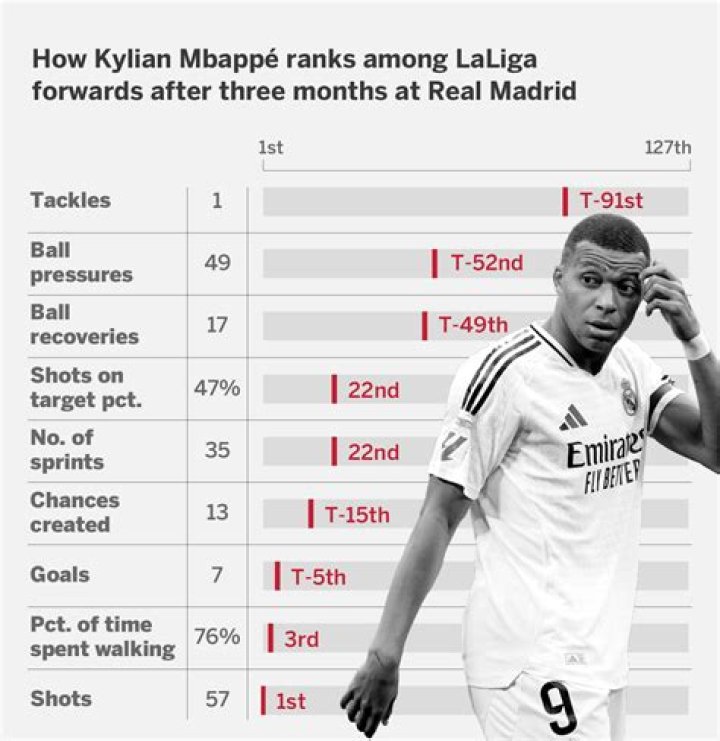
Is water an element or compound give reason?
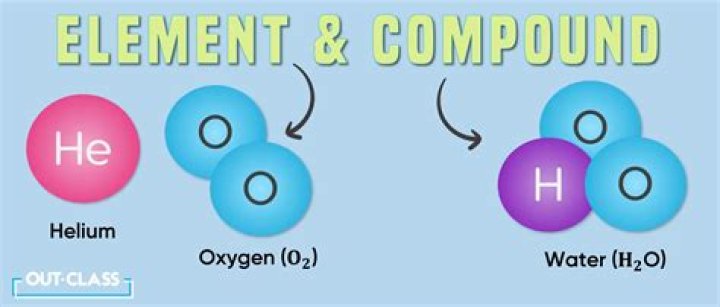
Thereof, is water an element or compound give reason in support of your answer?
Water is a compound because of the following reasons: It is composed of two different elements, hydrogen and oxygen which cannot be separated by physical methods. They can be separated only by electrolysis. The physical and chemical properties of hydrogen and oxygen are entirely different from the properties of water.
Also, what type of compound is water? Water (H2O) can be called a molecule or a compound because it is made of hydrogen (H) and oxygen (O) atoms. There are two main types of chemical bonds that hold atoms together: covalent and ionic/electrovalent bonds. Atoms that share electrons in a chemical bond have covalent bonds.
Beside this, is pure water an element?
Water, H2O, is a pure substance, a compound made of hydrogen and oxygen. Although water is the most abundant substance on earth, it is rarely found naturally in its pure form. Most of the time, pure water has to be created. Pure water is called distilled water or deionized water.
Is milk a compound?
Answer and Explanation: Milk is a mixture. Milk is not an element that is listed on the periodic table. Milk is not a single compound, but a mixture of compounds. The main
Related Question Answers
Is gold a compound?
Gold occurs in nature in both its native state and in compounds. The native state of an element is its free state. It is not combined with any other element. The most common compounds of gold are the tellurides.Who proved water is a compound?
Antoine LavoisierIs steel a compound?
Steel is a mixture of iron and carbon fused together with one or more other metals or nonmetals. Because steel is a mixture rather than a chemical compound, steel does not have a set chemical compound formula.Is Oxygen an element?
Element No. 8 on the Periodic Table of the Elements is a colorless gas that makes up 21 percent of Earth's atmosphere. Because it's all around, oxygen is easy to dismiss as dull and inert; in fact, it's the most reactive of the non-metallic elements.Is water a mixture?
A mixture is when two or more substances combine physically together. However, in water, two hydrogen atoms combine with one oxygen atom chemically, forming a new substance that has properties different from hydrogen alone or oxygen alone. Therefore, water is not a mixture; it is a compound and it is pure.Is h2o a element?
Originally Answered: is water a compound or an element? Yes, water is a compound. A compound forms whenever two or more atoms form chemical bonds with each other. The chemical formula for water is H2O, which means each molecule of water consists of one oxygen atom chemically bonded to two hydrogen atoms.How do you distinguish between a compound and a mixture?
No. Compound are substances which are formed by chemically combining two or more elements. Mixtures are substances that are formed by physically mixing two or more substances. Compounds are three types which are covalent compounds, metallic compounds and ionic compounds.Is oxygen a pure element?
In its naturally occurring state, oxygen is both an element and a molecule, but it is not a compound. It is an atomically homogeneous substance, meaning that there aren't any other kinds of atoms in an oxygen molecule. That makes it a pure element.What is pure water called?
Pure water, also known as purified water, is water from a source that has removed all impurities. Distilled water is the most common form of pure water. Pure water can be purified by carbon filtration, micro-porous filtration and ultraviolet oxidation. Some places use a combination of purification processes.Is gold a mixture?
Pure gold is an element. That is, it is not a combination of other materials. However, gold used in jewelry is usually a “mixture”, called an alloy mixed with other metals to harden it, to change the color as in copper makes the pink or rose gold that is currently enjoying great polarity.Is it safe to drink pure water?
Pure water, that is. Stripping water down to an ultrapure state makes it unfit for human consumption. In the world of electronics, manufacturers remove all of the minerals, dissolved gas and dirt particles from water. But if you were to drink ultra-pure water, it would literally drink you back.What is the purest substance on earth?
Ash is the purest thing in the world.What makes water pure?
What Is Distilled Water? Distilled water is created through the process of distillation. Basically, in the process of distillation, the pure H2O is boiled out of its contaminants. So, many of the contaminants found in water are inorganic minerals, metals etc.Can you make pure water?
Here's the thing, though: Pure water doesn't exist. Or, at the very least, it's not really possible on Earth. That limits humans' ability to generate pure water, because at a certain point it will start to dissolve the walls of its container.What are the 4 types of mixtures?
Within the categories of homogeneous and heterogeneous mixtures there are more specific types of mixtures including solutions, alloys, suspensions, and colloids. A solution is a mixture where one of the substances dissolves in the other. The substance that dissolves is called the solute.Is sugar and pure water a mixture?
Constant composition means that all parts of the mixture are the same. For example, dissolve sugar in water and mix it completely. Now take several samples from random areas. They will be the same, therefore this is a homogeneous mixture.What are the 4 types of compounds?
There are four types of compounds, depending on how the constituent atoms are held together:- molecules held together by covalent bonds.
- ionic compounds held together by ionic bonds.
- intermetallic compounds held together by metallic bonds.
- certain complexes held together by coordinate covalent bonds.
What are 5 examples of compounds?
Examples of compounds :- H20 = water.
- C6H12O6 = sugar.
- NaCl = salt.
- C2H6O = alcohol.
- C4H10= butane.
- NaHCO3 = baking soda.
- N20 = nitrogen.
- CH4 = methane.
What is the chemical name for h2o?
Water OxidaneWhat is the difference between elements and molecules?
An element has the same number of protons in its atoms regardless of its chemical state. A molecule is how an element or compound exists in a particular chemical system. The molecule is identified by chemical and physical measurements of a substance's properties.Is Salt a mixture?
A pure substance consists of a single element or compound. Iron is formed only of iron (Fe) atoms; table salt is formed only by sodium chloride (NaCl) molecules. A mixture, however, is made up of different compounds and/or elements. When salt is added to water to make saltwater, it becomes a mixture.What are examples of compounds?
Examples of compounds :- H20 = water.
- C6H12O6 = sugar.
- NaCl = salt.
- C2H6O = alcohol.
- C4H10= butane.
- NaHCO3 = baking soda.
- N20 = nitrogen.
- CH4 = methane.