The Jones Reagent is a mixture of chromic trioxide or sodium dichromate in diluted sulfuric acid, which forms chromic acid in situ. Although the reagent is very acidic, the substrate in acetone is essentially titrated with the oxidant solution and only very acid-sensitive groups are incompatible. Just so, is Jones reagent same as chromic acid?
The Jones Reagent is a mixture of chromic trioxide or sodium dichromate in diluted sulfuric acid, which forms chromic acid in situ.
Subsequently, question is, what does chromic acid do as a reagent? Chromic acid, H2CrO4, is a strong acid and a reagent for oxidizing alcohols to ketones and carboxylic acids.
Subsequently, one may also ask, which is Jones reagent?
The Jones Reagent is a solution of chromium trioxide in diluted sulfuric acid that can be used safely for oxidations of organic substrates in acetone. The reagent can also be prepared from sodium dichromate and potassium dichromate.
What is the formula of Jones reagent?
Jones' reagent | C3H8CrO8S - PubChem.
Related Question Answers
What are Jones and Collins reagents?
It is named after its discoverer, Sir Ewart Jones. The reaction was an early method for the oxidation of alcohols. Its use has subsided because milder, more selective reagents have been developed, e.g. Collins reagent. Jones reagent is a solution prepared by dissolving chromium trioxide in aqueous sulfuric acid. Which is called chromic acid?
CrO3​ is generally called chromic acid. Chromium trioxide CrO3​ is the acidic anhydride of chromic acid. Is chromic acid a carboxylic acid?
One of its hydroxyl groups is oxidized by way of a chromic half-ester in the same manner as alcohols. The resulting compound retains the hydroxyl group of the original chromic half-ester and is a carboxylic acid. Is Jones reagent selective?
Conclusions. Oxidation of benzyl and secondary alcohols using Jones reagent supported on graphite at room temperature under heterogeneous is a new and selective procedure for the preparation of corresponding benzaldehydes and ketones, respectively. All the substrates are commercially available. What is chromic acid test?
The chromic acid test uses the Jones reactant to oxidize aldehydes and alcohols and reduce the chromic acid, resulting in a color change. It is able to identify aldehydes, primary alcohol, and secondary alcohol. The Jones reactant is a mixture of chromium trioxide, sulfuric acid, and acetone. What is the Jones test?
The Jones dye test is used to assess patency of the lacrimal drainage system. In the first part of the test, a drop of fluorescein is placed in the conjunctival cul-de-sac. After 5 minutes, the nose is examined for the presence of dye. Is chromic acid a catalyst?
Alternative reagentsIn oxidations of alcohols or aldehydes into carboxylic acids, chromic acid is one of several reagents, including several that are catalytic.
How is Jones reagent different from Collins reagent?
As the Collins Reagent does not contain water (compared to the Jones Reagent) and is not as hygroscopic as is the Sarett Reagent, the oxidant is especially useful for the oxidation of primary alcohols to aldehydes where traces of water can lead to overoxidation. What is Jones oxidation test?
The Jones test utilizes chromium trioxide in the presence of sulfuric acid to act as a powerful oxidizing agent. In the presence of the Jones' reagent, a primary alcohol is first converted into an aldehyde and then into a carboxylic acid, while a secondary alcohol will be oxidized into a ketone. What is the reagent used in Jones oxidation?
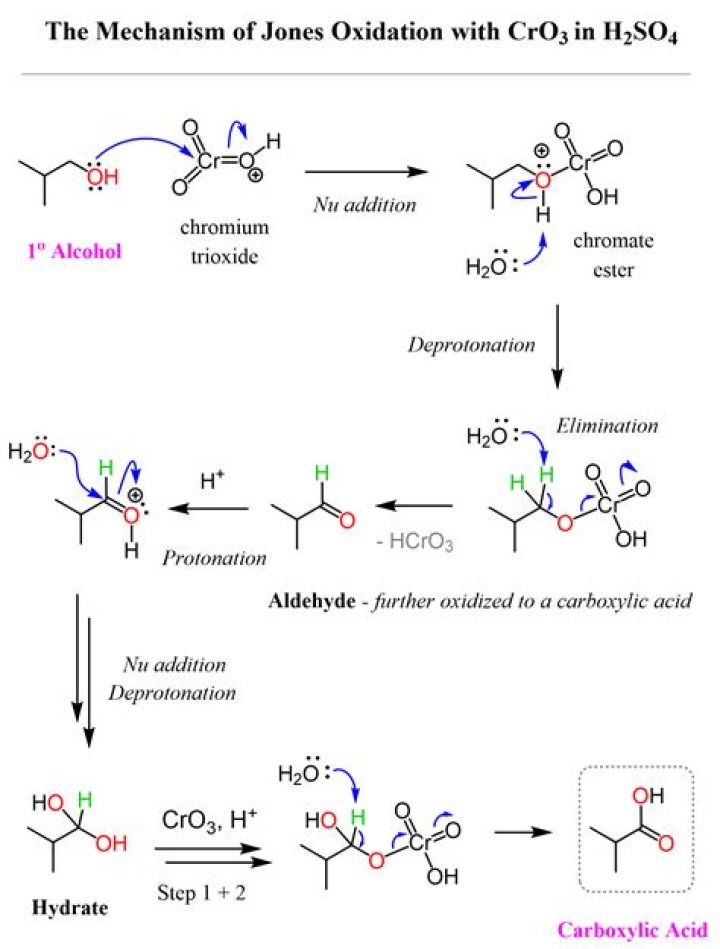
6.2.Secondary alcohols are oxidized to ketones, and primary alcohols can be oxidized to either an aldehyde or a carboxylic acid. This chromium trioxide/acetone/sulfuric acid reagent is often referred to as the Jones reagent, and oxidation of alcohols with this reagent is called Jones oxidation.
How does Jones reagent work?
Jones reagent oxidizes a secondary alcohol (such as 2-propanol) to the corresponding ketone (acetone). Jones reagent is aqueous and strongly acidic. Jones reagent oxidizes a primary alcohol to an aldehyde, and subsequently to a carboxylic acid. Is PCC a Jones reagent?
Both Jones reagent and PCC turn a secondary alcohol into a ketone, going from C-OH to C=O. PCC turns a primary alcohol into an aldehyde (C-OH to C=O). However, Jones reagent is strong and oxidizes primary alcohol further to carboxylic acids (C-OH to COOH). Is Jones reagent same as PCC?
PCC is a milder version of the Jones reagent. Oxidation with PCC converts 1° alcohols to aldehydes: RCHâ‚‚OH → RCHO. The Jones reagent continues the oxidation to the carboxylic acid: RCHâ‚‚OH → [RCHO] → RCOOH. Is Jones reagent a strong oxidising agent?
But this says that it is an extremely strong oxidising agent and can oxidise Aldehydes to Carboxylic Acids too (thus giving acids on the oxidation of 1∘ alcohols). How is chromic acid reagent prepared?
Chromic acid is a commonly used glassware cleaning reagent. It is prepared in a one liter container by dissolving 60 grams of potassium dichromate in approximately 150 mls of warm distilled water and then slowly adding concentrated sulfuric acid to produce a total volume of one liter Chromic Acid solution. How do you test for chromic acid?
Three drops of the compound to be tested are mixed with 5 drops of acetone and 5 drops of chromic acid solution (an orange solution). Indications of a positive test: The disappearance of the red-orange color of chromic acid and the formation of a blue-green color of the Cr (III) ion indicates a positive test. Is chromic acid organic or inorganic?
2. Any organic product formed whose molecule has at least one hydrogen atom bonded to the carbonyl carbon is further oxidized by chromic acid. Oxidation of a primary alcohol by chromic acid results in a carboxylic acid as the organic product. What is chromic acid mixture?
Chromic acid is made by mixing Potassium dichromate and an acid such as Hydrochloric acid or Sulfuric acid. It is an extremely strong oxidizing agent that will react violently with any organic compound. What is the anion for chromic acid?
The ''chromic'' in chromic acid comes from the polyatomic ion chromate. A polyatomic ion is a molecule with an electric charge. Chromate has a charge of negative two with the formula CrO4 -2. Is chromic acid the same as hexavalent chromium?
Hexavalent chromium [Cr(VI)] is one of the valence states (+6) of the element chromium. The Cr(VI) compound chromic acid is used to electroplate chromium onto metal parts to provide a decorative or protective coating. Can Jones reagent oxidize aldehydes?
Primary and secondary alcohols are oxidized to aldehydes ketones using Jones reagent, Collins oxidation, PCC, or PDC. Primary alcohols are oxidized to aldehydes and secondary alcohols to ketones by Swern oxidation. Is chromic acid soluble in water?
Water Alcohol What does the Jones reagent look like?
* The Jones reagent is a mixture of chromic anhydride and dilute sulfuric acid (CrO3 + H2SO4 + H2O) in acetone. It is used in the oxidation of secondary alcohols, that do not contain acid sensitive groups, to corresponding ketones. How Jones reagent is differ from other chromium reagents like PCC PDC?
Oxidation with the PCC reagent converts 1º-alcohols to aldehydes; whereas Jones reagent continues the oxidation to the carboxylic acid product, as shown in the second reaction. Is tollens a reagent?
Tollens' reagent is an alkaline solution of ammoniacal silver nitrate and is used to test for aldehydes. Silver ions in the presence of hydroxide ions come out of solution as a brown precipitate of silver(I) oxide, Ag2O(s). This precipitate dissolves in aqueous ammonia, forming the diamminesilver(I) ion, [Ag(NH3)2]+. Is Lucas a reagent?
Lucas' reagent is a solution of anhydrous zinc chloride in concentrated hydrochloric acid. This solution is used to classify alcohols of low molecular weight. The reaction is a substitution in which the chloride replaces a hydroxyl group. Which of the following is sarett reagent?
The mild oxidation of primary or secondary alcohols into corresponding aldehydes or ketones using chromium trioxide and pyridine complex as the oxidant is generally referred to as the Sarett oxidation. The complex from anhydrous chromium trioxide and pyridine is commonly termed the Sarett's reagent. What does SOCl2 do in a reaction?
Carboxylic acids react with Thionyl Chloride (SOCl2) to form acid chlorides. During the reaction the hydroxyl group of the carboxylic acid is converted to a chlorosulfite intermediate making it a better leaving group. The chloride anion produced during the reaction acts a nucleophile. What is meant by tollens reagent?
Tollens' reagent (chemical formula. ) is a chemical reagent used to distinguish between aldehydes and ketone functional groups along with some alpha-hydroxy ketones which can tautomerize into aldehydes.