How much power does an Aluminium smelter use?
In this regard, how much electricity does an Aluminium smelter use?
It is estimated that the cost of electricity for the smelter is $14 per MWh (1.4 cents per kWh), and that it has cost the state more than $2 billion over 20 years. The Portland smelter is believed to consume between 8 and 10 per cent of the state's electricity.
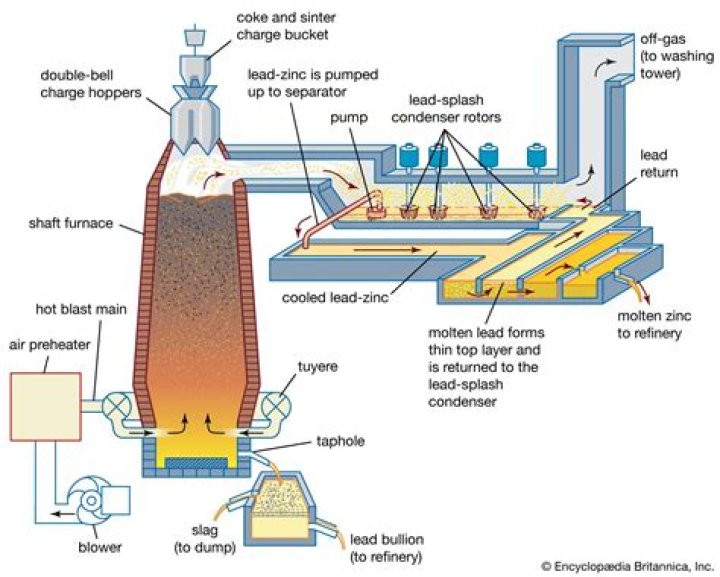
Secondly, how does an Aluminium smelter work? The steps in aluminum smelting are described below: Alumina is dissolved in molten cryolite at 1,000 degrees C (1,832 degrees F). At the cathode, electrolysis reduces aluminum ions to aluminum metal. At the anode, carbon is oxidized to form carbon dioxide gas.
Also, how much power does tiwai use?
Electricity use The smelter's power demand from the national grid is about 570 MW. Most of the energy for the smelter is supplied from the Manapouri hydroelectric power station, via two double circuit 220 kV transmission lines.
Why does the extraction of Aluminium require so much energy?
Extraction of aluminium It is expensive, largely because of the amount of electricity required in the extraction process. The ions in the aluminium oxide must be free to move so that electricity can pass through it. Aluminium oxide has a very high melting point (over 2000°C) so it would be expensive to melt it.
Related Question Answers
Which country is the largest producer of Aluminium?
ChinaDoes aluminum lose strength over time?
Just like steel, aluminum alloys become weaker as the service temperature rises. But aluminum melts at only about 1,260 degrees, so it loses about half of its strength by the time it reaches 600 degrees.What forms of energy are needed for processing aluminum?
The aluminum production process uses electrical power as major energy source. This electrical energy is generally derived from thermal power plants which work with maximum 30% efficiency.How much does it cost to extract and process aluminum?
The total cost per tonne to extract Aluminium is $1,632.Is aluminum expensive to produce?
Like many metals, aluminium does not come out of the ground as a shiny metal, but as a special kind of rock, an ore. The production of aluminium is very expensive and extremely damaging to the environment. To make aluminium the ore (bauxite) must first be mined.Why does the extraction of Aluminium use so much energy?
It is expensive, largely because of the amount of electricity required in the extraction process. Aluminium ore is called bauxite . This is an aluminium compound with a lower melting point than aluminium oxide. The use of cryolite reduces some of the energy costs involved in extracting aluminium.How is Aluminium smelted?
Aluminium smelting is the process of extracting aluminium from its oxide, alumina, generally by the Hall-Héroult process. Alumina is extracted from the ore bauxite by means of the Bayer process at an alumina refinery. Smelters are often located near ports, since many smelters use imported alumina.How is Aluminium made step by step?
It takes about 4 lb (2 kg) of bauxite to produce 1 lb (0.5 kg) of aluminum metal. Aluminum is manufactured in two phases: the Bayer process of refining the bauxite ore to obtain aluminum oxide, and the Hall-Heroult process of smelting the aluminum oxide to release pure aluminum. of aluminum.What is Tiwai Point?
The Tiwai Point Aluminium Smelter is an aluminium smelter owned by Rio Tinto Group (79.36%) and the Sumitomo Group (20.64%), via a joint venture called New Zealand Aluminium Smelters Limited. The facility, New Zealand's only aluminium smelter, is at Tiwai Point, near Bluff.How is bauxite ore mined?
Mining bauxite Bauxite is usually found near the surface of terrain and can be strip-mined economically. When the land is cleared prior to mining, the topsoil is stored so it can be replaced during rehabilitation. During the strip-mining process, bauxite is broken up and taken out of the mine to an alumina refinery.How is Aluminium made in NZ?
Operations. The smelter uses the Hall-Héroult process to convert alumina (aluminium oxide) to elemental aluminium. Bauxite is mined in Australia and refined into alumina before being shipped to New Zealand. In 2011 the smelter produced 354,030 saleable tonnes of aluminium, which was its highest ever output at the time.Why is Aluminium so useful?
The metal prevents oxygen, moisture, and bacteria from damaging the food. Pharmaceuticals use aluminium in their packaging as well. This is because it can store perishable goods without needing refrigeration. The way you look at it, aluminium is the superhero of all metals.How do you melt aluminum at home?
Melting the Aluminum- The first step you'll want to take is to crush the cans so that you can load as many as possible into the crucible.
- Fire up the kiln or furnace to 1220°F.
- Put on safety glasses and heat-resistant gloves.
- Open the kiln.
- Pour the liquid aluminum into the mold.