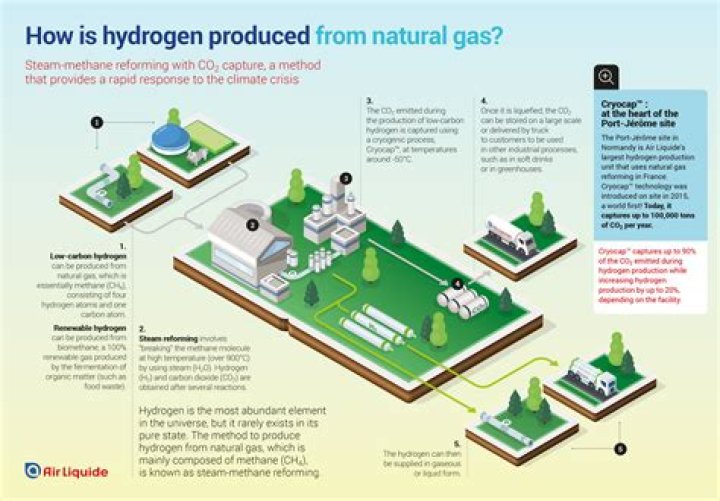
There are a number of ways to produce hydrogen: - Natural Gas Reforming/Gasification: Synthesis gas, a mixture of hydrogen, carbon monoxide, and a small amount of carbon dioxide, is created by reacting natural gas with high-temperature steam.
- Electrolysis: An electric current splits water into hydrogen and oxygen.
Considering this, what is the best way to produce hydrogen?
The two most common methods for producing hydrogen are steam-methane reforming and electrolysis (splitting water with electricity.
Beside above, can you make your own hydrogen fuel? Yes, it's possible to generate hydrogen in a science fair kind of way by electrolysing water. A liter of water will get you about 111 grams of hydrogen if you can capture it all. A kilogram of hydrogen is the fuel cell car equivalent to a gallon of gas.
Considering this, how is hydrogen made?
Hydrogen fuel can be produced from methane or by electrolysis of water. In electrolysis, electricity is run through water to separate the hydrogen and oxygen atoms. This method can use wind, solar, geothermal, hydro, fossil fuels, biomass, nuclear, and many other energy sources.
Does hydrogen come from natural gas?
Currently, most hydrogen is produced from fossil fuels, specifically natural gas. Electricity—from the grid or from renewable sources such as wind, solar, geothermal, or biomass—is also currently used to produce hydrogen.
Related Question Answers
What is the cheapest way to produce hydrogen?
Steam reforming is a hydrogen production process from natural gas. This method is currently the cheapest source of industrial hydrogen. The process consists of heating the gas to between 700–1100 °C in the presence of steam and a nickel catalyst. How hard is it to produce hydrogen?
Hydrogen is an important, energetic element, which is used in a variety of applications ranging from making semiconductors to powering fuel cells. However, it's also a difficult, expensive one to produce in pure molecular form. Is hydrogen expensive to produce?
The cost of hydrogen production is an important issue. Hydrogen produced by steam reformation costs approximately three times the cost of natural gas per unit of energy produced. This means that if natural gas costs $6/million BTU, then hydrogen will be $18/million BTU. What is the main problem in using hydrogen as fuel for vehicles?
The only real problem is the issue of safety. Hydrogen is highly flammable – more so than regular fuel – and is harder to contain than oil. This means that any car fitted with hydrogen fuel cells is a lot more likely to blow up or set on fire if it is exposed to extreme heat, if it breaks down or if it overheats. How dangerous is hydrogen?
Physical dangers: The gas mixes well with air, explosive mixtures are easily formed. The gas is lighter than air. Chemical dangers: Heating may cause violent combustion or explosion. Reacts violently with air, oxygen, halogens and strong oxidants causing fire and explosion hazard. Is hydrogen easy to produce?
Hydrogen is easily produced from water by electrolysis, a process which uses electricity to break the bonds between water's constituent elements, hydrogen and oxygen, and releases them as gas. Currently, industrial production of hydrogen relies overwhelmingly on fossil fuels to power the electrolysis process. How much electricity does it take to produce hydrogen?
A 100% efficient electrolyser requires 39 kWh of electricity to produce 1 kg of hydrogen. The devices today require as much as 48 kWh/kg. So, if electricity costs are 0.05 US$/kWh, the power cost for the electrolysis process alone is 2.40 US$/kg of hydrogen. What are the two most common ways to produce hydrogen gas used in fuel cells?
The two most common ways to produce hydrogen are steam reforming (using high-temperature steam to produce hydrogen from natural gas) and electrolysis (splitting water). Why hydrogen is not used as a domestic fuel?
When hydrogen comes in contact with air, it reacts explosively. Hydrogen needs to be compressed at higher pressures. Therefore, though hydrogen gas has the highest calorific value, it cannot be used as a fuel. Does hydrogen burn with a blue flame?
Hydrogen burns with a pale blue flame that is nearly invisible in daylight. A pure hydrogen flame will not produce smoke. Hydrogen flames have low radiant heat. How much does a gallon of hydrogen fuel cost?
$0.0015/gallon + $0.987/kg (gge) Refining Costs = $0.9885 = $1.00/kg (gge) using Atmospheric Electrolyses. ÷ 50 gallons per drum = 6.66 “barrels of H2” per 1000 gallons of water. That is equivalent to a 7:1 H2 to crude oil ratio. Energy needed to make 1 kg of H2 = 32.9 kWh/kg. Is hydrogen fuel cheaper than gasoline?
While hydrogen is a cheaper fuel than gasoline on paper, the reality is, as of 2010, it is much more expensive. The few models of hydrogen-fueled cars that are commercially available generally cost more than $100,000. Thus, fueling stations are limited, making the cost of hydrogen fuel vary widely. Is hydrogen fuel liquid or gas?
LIQUEFACTION: Chilled to near absolute zero, hydrogen gas turns into a liquid containing one-quarter the energy in an equivalent volume of gasoline. The technology is well-proven: For decades, NASA has used liquid hydrogen to power vehicles such as the space shuttle. Are there any hydrogen powered cars?
As of 2019, there are three models of hydrogen cars publicly available in select markets: the Toyota Mirai which is the world's first mass produced dedicated fuel cell electric vehicle, the Hyundai Nexo, and the Honda Clarity. Where is hydrogen found?
Hydrogen is easily the most abundant element in the universe. It is found in the sun and most of the stars, and the planet Jupiter is composed mostly of hydrogen. On Earth, hydrogen is found in the greatest quantities as water. Is hydrogen fuel the future?
Hydrogen is the fuel of the future. Hydrogen is an energy carrier that can be used in internal combustion engines or fuel cells producing virtually no greenhouse gas emissions when combusted with oxygen. The only significant emission is water vapor. Why is it difficult to use hydrogen as a source of energy?
=> it burns with an explosion.. So ,it's use is very much unsafe. =>It is very expensive to produce hydrogen from electrolysis of water. =>It is very difficult to store and transport hydrogen because it is explosively inflamable. Why are hydrogen engines a bad idea?
Hydrogen fuel cells have bad theoretical and practical efficiency. Hydrogen storage is inefficient, energetically, volumetrically and with respect to weight. It has a horrible well-to-wheel efficiency as a result. Easy ways to get large quantities of hydrogen are not 'cleaner' than gasoline. Can hydrogen fuel cells explode?
According to OSHA, “Hydrogen used in the fuel cells is a very flammable gas and can cause fires and explosions if it is not handled properly. How many MPG does a hydrogen car get?
70 miles per gallon
Does hydrogen water really work?
Hydrogen water is said to increase energy, reduce inflammation, and reduce recovery times after workouts. A study of 10 soccer players showed that drinking hydrogen water may reduce muscle fatigue and muscle function decline caused by exercising. How do you make your own fuel?
Alcohol Fuel Basics. It takes some mechanical aptitude, but you can make your own fuel by fermenting appropriate feed stocks into 96 proof alcohol. For small producers, the best choices as ethanol feedstocks are crops with a lot of starch or sugar, such as corn (shown above) or sugar beets. Why hydrogen is not used in cars?
The reason why hydrogen is inefficient is because the energy must move from wire to gas to wire in order to power a car. This is sometimes called the energy vector transition. Let's take 100 watts of electricity produced by a renewable source such as a wind turbine. How much does a hydrogen tank cost?
If you have a particular hydrogen tank in mind, you can enter its size and cost. Click the Click to add new item option to add more rows to the table. In the example above, a 1 kg tank costs $1,400 initially, $1,200 to replace at the end of its life, and $30/yr for operation and maintenance. How can water be split without electricity?
More than 352 thermochemical cycles have been described for water splitting or thermolysis., These cycles promise to produce hydrogen oxygen from water and heat without using electricity. Since all the input energy for such processes is heat, they can be more efficient than high-temperature electrolysis. Can engine run on salt water?
The 920 horsepower (680 kW) QUANT e-Sportslimousine uses an electrolyte flow cell power system to propel the four electric motors within the car. Using the same principles as a hydrogen fuel cell, the liquid used for storing energy is, astonishingly, saltwater. Is natural gas renewable or nonrenewable?
Natural gas is one non-renewable energy source. What is the source of hydrogen for plants?
Hydrogen (H), derived almost entirely from water, is one of the 17 essential nutrients necessary for plant growth. Is hydrogen a gas at room temperature?
Hydrogen is a gas at room temperature. Elemental hydrogen (H, element 1), nitrogen (N, element 7), oxygen (O, element 8), fluorine (F, element 9), and chlorine (Cl, element 17) are all gases at room temperature, and are found as diatomic molecules. Can hydrogen fuel become reliable renewable energy?
Use of Hydrogen fuel modular power plants away from residential areas along road side to feed EVs via overhead supply line, will be safe from hazard and work as renewable energy source for saving Climate.