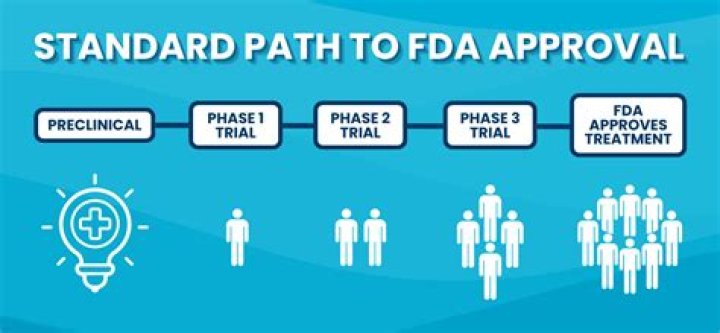
How do I change FDA regulations?
Besides, can the president change FDA regulations?
The short answer is no. Though Congress granted Health and Human Services broad discretion to issue EUAs, it also established clear standards, requiring that emergency authorizations be issued only on the basis of “scientific evidence” and for the purpose of protecting public health.
Subsequently, question is, how does the FDA create regulations? FDA develops regulations based on the laws set forth in the Food, Drug, and Cosmetic Act (FD&C Act) or other laws – including the Family Smoking Prevention and Tobacco Control Act – under which FDA operates. FDA regulations have the full force of law.
Beside this, are FDA regulations legally binding?
No. The FDA guidance documents are not legally binding on the public or the FDA. BUT, the FDA has come to rely on guidance documents as a means of informal policy making.
How do you comment on proposed regulations?
To read the Federal Register online, go to gpoaccess.gov. Once you have identified the proposed rule you would like to comment on, you will need to locate the rule on Regulations.gov, a website run by the federal government, designed to allow the public to find proposed and final regulations and to comment on them.
Related Question Answers
Who does FDA report to?
FDA is an agency within the Department of Health and Human Services.Who has authority over FDA?
United StatesWhat branch of government does the FDA fall under?
Executive branchWho is over the president?
Present line of succession| No. | Office | Party |
|---|---|---|
| 1 | Vice President | Democratic |
| 2 | Speaker of the House of Representatives | Democratic |
| 3 | President pro tempore of the Senate | Democratic |
| 4 | Secretary of State | Democratic |
What legislative power does the FDA have?
FDA has the power to suspend or revoke a license. Administratively, FDA can do that itself; it does not have to go to court. And, FDA has the power to recall products. SLIDE 22 Under the PHS Act, in addition to the licensing provisions, FDA can also take actions to prevent communicable diseases.What are FDA regulations?
FDA's traditional "safe and effective" standard for evaluating medical products does not apply to tobacco products. FDA regulations are based on the laws set forth in the Tobacco Control Act and the Food, Drug, and Cosmetic Act (FD&C Act). FDA regulations are also federal laws.What is President in charge of?
The President is responsible for implementing and enforcing the laws written by Congress and, to that end, appoints the heads of the federal agencies, including the Cabinet. The Vice President is also part of the Executive Branch, ready to assume the Presidency should the need arise.Which consumer protection laws is the FDA tasked with enforcing?
Under the Federal Food, Drug, and Cosmetic Act (the Act), FDA has the broad mandate to assure safety and effectiveness of drugs (including animal drugs), devices (including veterinary devices), and the safety of the food supply.Why do we need FDA regulations?
The mission of FDA is to enforce laws enacted by the U.S. Congress and regulations established by the Agency to protect the consumer's health, safety, and pocketbook.What is FDA final rule?
The Food and Drug Administration (FDA) is issuing a final rule to establish postmarketing safety reporting requirements for combination products. Finally, the final rule requires that constituent part applicants share certain postmarketing safety information with each other.Why are the food regulations important?
Food regulations are important, and necessary because they enforce rules so that the buyer stays safe, and that the health of the consumer is protected. It ensures the wholesomeness quality.What is the difference between laws regulations and guidance?
While regulations cover a topic broadly, rules get into the nitty-gritty. As part of a regulation, they have the force of law. Guidance. Guidance is supplemental material published by an agency that helps clarify existing rules.Who controls EPA regulations?
The current Administrator is Michael S. Regan. The EPA is not a Cabinet department, but the administrator is normally given cabinet rank. The EPA has its headquarters in Washington, D.C., regional offices for each of the agency's ten regions, and 27 laboratories.What consumer rights does the FDA protect?
The FDA protects the public from unsafe foods to drugs and from medical devices to cosmetics. It also protects the rights and safety of patients in clinical trials of new medical products and monitors the promotional activities of drug and device manufacturers.What is difference between FDA Usfda?
USFDA: The Food and Drug Administration (FDA) is an agency within the U.S. Department of Health and Human Services. FDA's responsibilities extend to the 50 United States, the District of Columbia, Puerto Rico, Guam, the Virgin Islands, American Samoa, and other U.S. territories and possessions.Who made rules and regulations?
The United States Congress is the lawmaking body of the Federal Government. Congress has two houses: the House of Representatives and the Senate. Each state also passes its own laws, which you must follow when you are in that state.Where can I find FDA regulations?
CFR Resources:- 21 CFR Database on FDA.gov. Searchable database; updated once a year.
- GPO Electronic Code of Federal Regulations (eCFR) Currently updated online version.
What is FDA compliance?
FDA compliant is a way of describing products and materials that are safe for direct contact with food. A Food Contact Substance is any material that comes into contact with or is used for manufacturing, product packing, packaging, transporting or for holding food.What is the rule making process?
In administrative law, rulemaking is the process that executive and independent agencies use to create, or promulgate, regulations. In general, legislatures first set broad policy mandates by passing statutes, then agencies create more detailed regulations through rulemaking.What is the FDA requirements for off site caterer?
The food can be properly stored and segregated to protect it from any source of contamination. Direct transport equipment need not be powered, but if hot and/or cold holding equipment is provided by the caterer for use at the site of the banquet, the equipment must operate under power.What is FDA declaration?
Recognition is the process whereby the FDA identifies standards to which manufacturers of medical devices may submit a declaration of conformity to demonstrate they have met relevant requirements in the Federal Food, Drug, and Cosmetic Act (FD&C Act). The FDA usually performs the activity at least twice annually.What is the meaning of 21 CFR?
Code of Federal RegulationsHow do you write an effective comment?
Top ten tips for writing a great comment- Read the article. It sounds obvious, but you'd be surprised how many comments can be answered with the words “it says in the article”.
- Respond to the article. …
- Read the other comments.
- Make it clear who you're replying to.
- Use the return key.
- Avoid sarcasm.
- Avoid unnecessary acronyms.
- Use facts.
How do you write an effective public comment?
Use an opening sentence to establish your credibility. State who you are and summarize any of your experiences that are relevant to the topic of the proposal. Use the next few sentences to succinctly summarize the data or research that you have provided.How do I submit a public comment?
The option to submit a digital public comment on a federal agency document can be done in 2 ways: either directly through the agency's site, or through the Federal Register. Both paths will lead to a document open for comments on regulations.gov, the website for the Federal Register.How do you write a legislative comment?
Following are the tips for making the comment clear, concise, and easy to follow. ? If one has more than one major concern, open the comment with a summary section like “Major Concerns” or “General Comments” and outline them before you give detail. ? Use headings and sub-headings to separate the major points.How do I comment on government regulations?
How do I submit a comment on Regulations.gov?- Docket Details page when a document within the docket is open for comment,
- Document Details page when the document is open for comment, and.
- Document Search Tab with all search results open for comment displaying a "Comment" button.
How do you use regulations government?
Submit an Electronic Comment- Go to regulations.gov and click on "Advanced Search" (right below the "Search" button).
- Type keyword(s) or Docket ID in the "Search" box.
- Select "Open for Comment" (directly under Search box).
- Select by Document Type "Notice," "Proposed Rule," and "Rule."